This Fox Chase professor participates in the Undergraduate Summer Research Fellowship.
Learn more about Research Volunteering.
Related Articles
00 / 00
-

-

The OCRA Liz Tilberis Early Career Award provides James Duncan, PhD, with a three-year grant of $150,000 per year for research on ovarian cancer
-

-

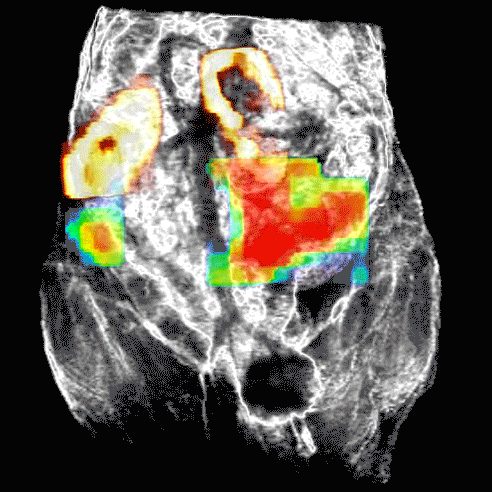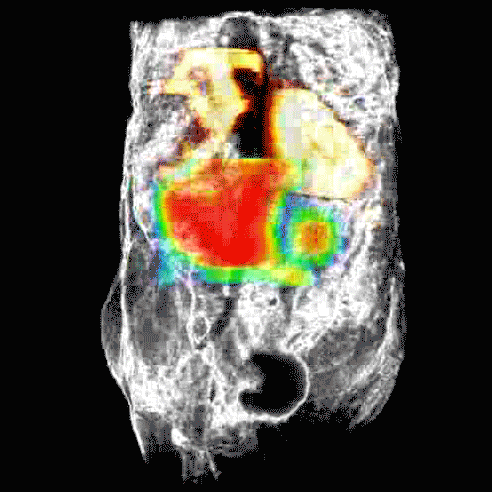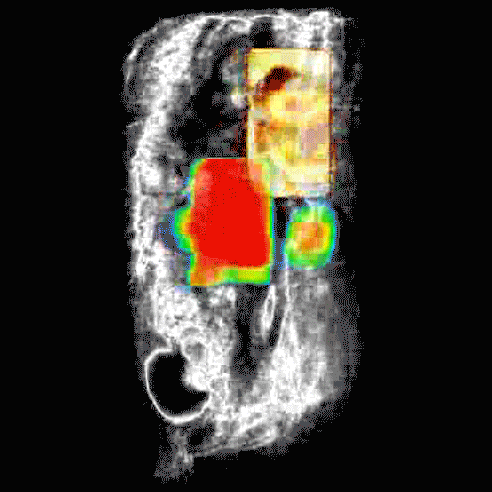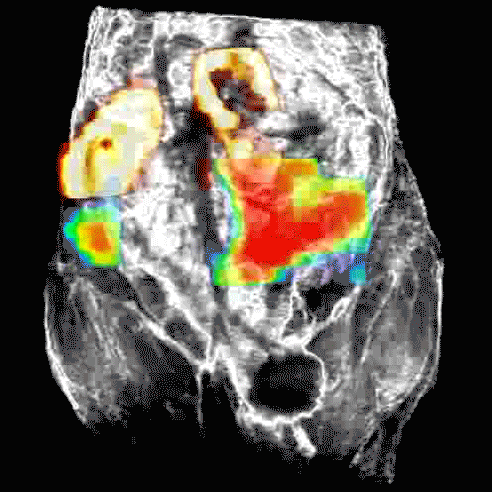
Disruption of HSP90 function can lead to death of ovarian cancer cell lines without known defects in DNA repair mechanisms, such as BRCA1/2 mutations, when used in combination with a PARP inhibitor.
-

Denise C. Connolly, PhD
-

A photograph of Denise Connolly standing in a cluttered laboratory, smiling at the camera.
-

Denise Connolly, PhD
-

-



















Patient comments